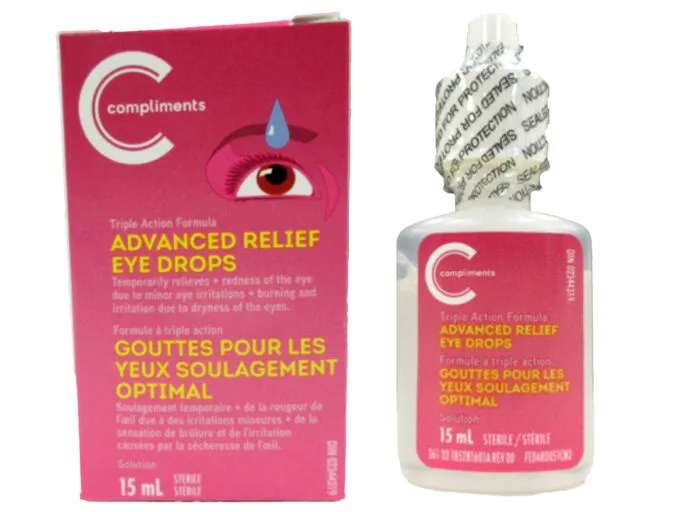
Advanced Relief Eye Drops Compliments Pharmasave Teva Canada Recall Overview:
- Who: Teva Canada Ltd. is recalling certain lots of Pharmasave Advanced Relief Eye Drops and Compliments Advanced Relief Eye Drops.
- Why: Some bottles may contain ingredients not listed on the label that may pose health risks to those allergic to them.
- Where: The recall is effective in Canada.
Teva Canada Ltd. is recalling certain lots of Pharmasave Advanced Relief Eye Drops and Compliments Advanced Relief Eye Drops after a customer complaint led the company to realize it had put the wrong solution in the bottles.
The Health Canada website posted the recall May 7. It applies to certain lots of Compliments Advanced Relief Eye Drops, 15 mL, and Pharmasave Advanced Relief Eye Drops, 15 mL, with expiry 31 March, 2024.
The products are used for the temporary relief of eye redness due to minor eye irritation, burning and dryness.
Following a complaint involving the Pharmasave eye drops, Teva Canada identified that some bottles may have been packaged with another eye drop product that contains different active ingredients.
The company is not currently facing legal action over the recall, but Top Class Actions follows recalls closely as they sometimes end in class action lawsuits.
Eye Drops Packaged in Incorrect Bottle, Teva Canada Says
Teva Canada mistakenly placed Naphazoline HCl and glycerine in bottles that were not meant to contain those ingredients, the company says.
The two ingredients may cause a reaction in individuals allergic to them. Signs of an allergic reaction include rash, itching or swelling, especially of the face, tongue and throat.
“Do not use the affected product. Consult a health care professional if you have used this product and have health concerns,” the recall states.
Cartons of the affected Pharmasave and Compliments eye drops are labeled with lot number AR21C03. The affected bottles may be labeled with either lot number AR21C03 or RL21D01.
In 2019, a Canadian class action lawsuit claimed that Lasik MD did not warn patients of the risk of permanent nerve damage associated with the popular laser eye surgery. The case was dismissed in 2020 with the judge ruling that claims weren’t brought in time.
What do you think of this recall? Let us know in the comments!
Don’t Miss Out!
Check out our list of Class Action Lawsuits and Class Action Settlements you may qualify to join!
Read About More Class Action Lawsuits & Class Action Settlements:
- TJX Canada Recalls Pimlico Confectioners, Keats London Vegan Chocolate Products Due To Undeclared Milk
- British Columbia, Insurance Company Face Class Action Over Illegal Medical Fee Reimbursement
- B.C. Judge Dismisses WestJet Class Action Over Travel Credits
- Families of Alleged MK Ultra Brainwashing Victims Protest in Montreal














3 thoughts onTeva Canada Recalls Advanced Relief Eye Drops Due To Packaging Error
I have used these drops from Pharmasave for years
Please add me to the list. I bought these eye drops and caused my eyes to swell and become really irritated.
I purchased these drops many times for me and for my mother. I thought I had seasonal allergies and my mother was treated for an actual eye disease that caused her eyelashes to fall out. Please advise of any further settlement details thank you